
NOUVEAU
Découvrez le PACK FAMILLE !
Le Pack Famille se compose de :
– 2 autotests COVID-VIRO ALL IN adapté à l’enfant
– 2 autotests COVID-VIRO
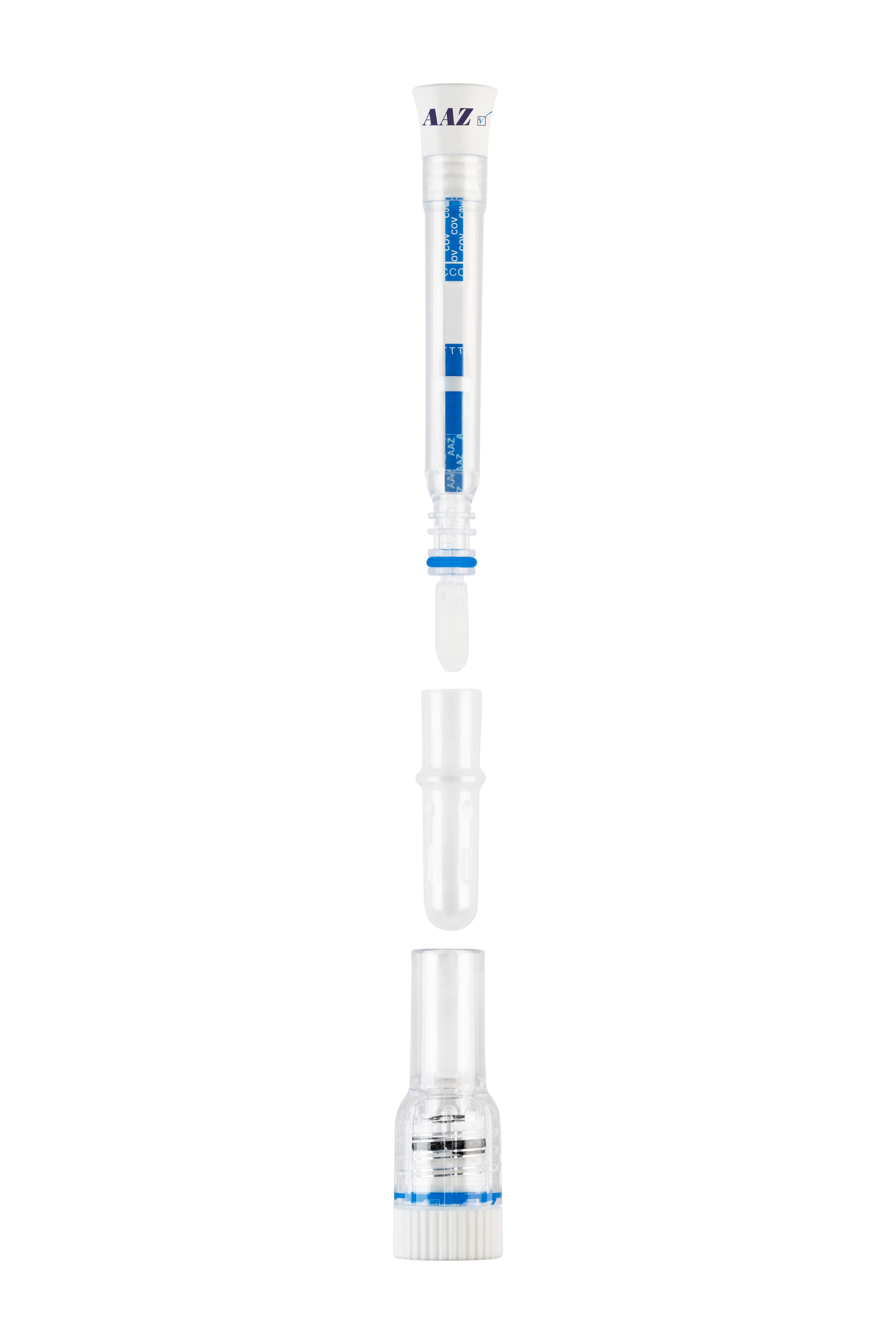
autotest COVID-VIRO ALL IN® is an in vitro diagnostic device for one-time use only.
autotest COVID-VIRO ALL IN® kit requires no special equipment other than what is included in the box. The test can be done at home or with the help of a healthcare professional.
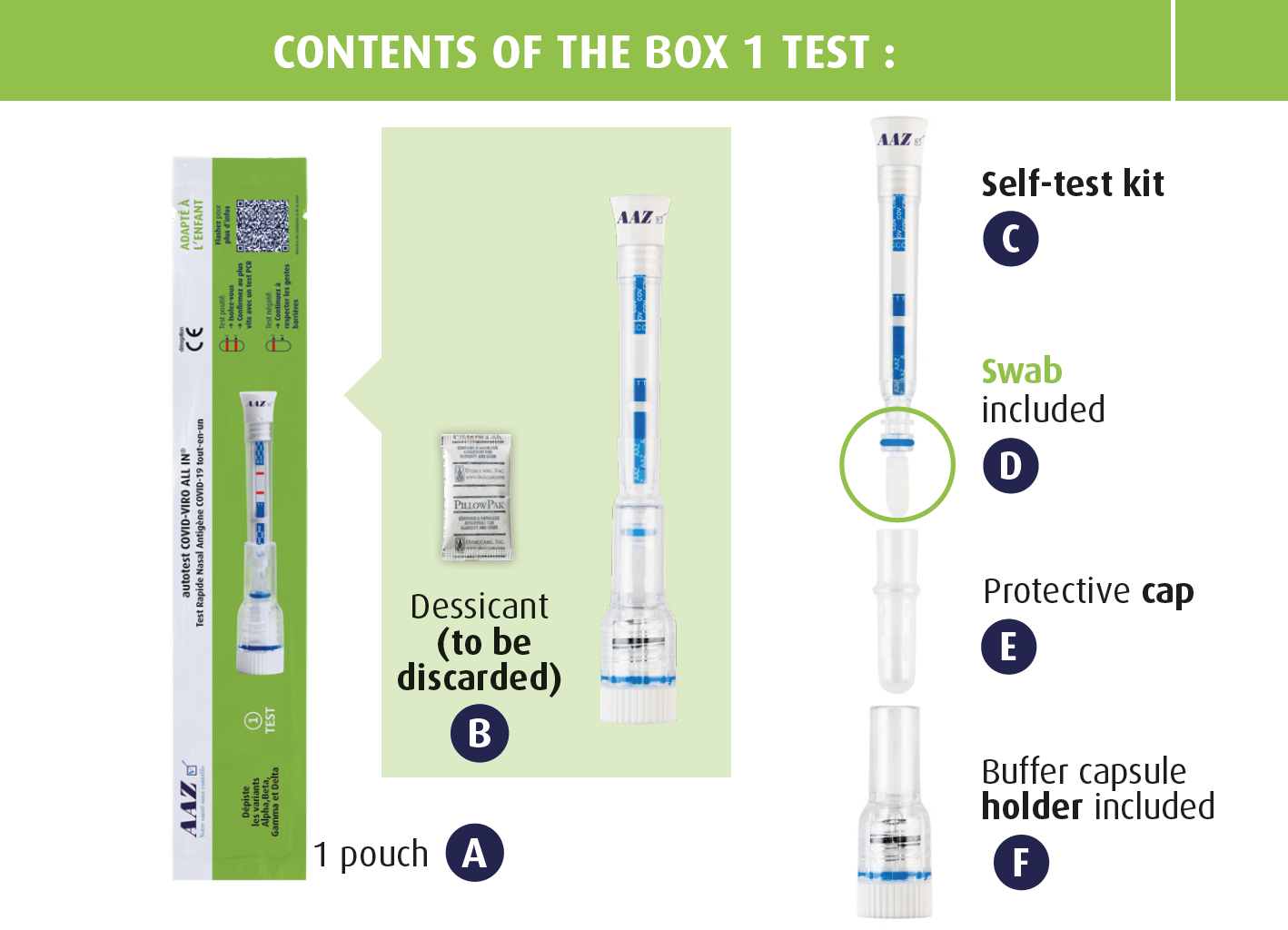
Inside the single-test box, you will find :
- illustrated instructions on how to do the test,
- 1 pouch containing 1 self-test.
Before you do the test, ensure the self-test kit is at room temperature (15-30°C), read the user manual and watch the demo video. It is easy to get and read the result from autotest COVID-VIRO ALL IN®. The result appears as lines in just 15 minutes.
If you have any doubts about your results, contact your doctor.
Packaging: 10 tests per box (AS-COV-005) or 1 test per box (AS-COV-007)
Storage conditions: between 2 and 30 °C
Sample type: nasal
Reliability: a comparative clinical study conducted at Orléans Regional Hospital showed a 97,5% correlation between results from autotest COVID-VIRO ALL IN® and those form the RT-PCR laboratory test.
Sensitivity: the sensitivity of this self-test has been estimated at 93,02% (with confidence intervals ranging from 81,4% to 97,6%), i.e. 6,98% flase negatives*.
Specificity: the specificity of this self-test has been estimated at 100% (with a confidence interval ranging from 95,2% to 100%). No false positives were identified**.
Usability: the usability study of this self-test by lay people showed that 98% of participants who use this self-test obtain an interpretable result and that 99% of self-tests are interpreted correctly.
Interference: examination of samples containing substances likely to distort the results of this test and from people with conditions likely to do so revealed no notable interference.
*False negative: a sample known to be positive for the target marker and wrongly classed as negative by the device.
**False positive: a sample known to be negative for the target marker and wrongly classed as positive by the device.
-
autotest COVID-VIRO ALL IN® is intended only for private use to detect the COVID-19 virus; under no circumstances should it be used for other diagnoses or other purposes.
-
Store the self-test in its original package in a cool and dry place between 2°C and 30°C.
-
If the box is stored in a refrigerator (between 2°C and 8°C), it must be brought to room temperature (between 15°C-30°C) before use.
-
The self-test is for single use only. Do no reuse it under any circumstances.
-
Do not open the packege until you are ready to do the test.
-
Do not use the self-test after the expiry date.
-
Do not use the cassette if the packaging is open or damaged.
-
Do not do the test in a room where there is a strong current of air, such as from a fan or powerful air-conditioning.
-
Storing the test in conditions other that those stated, or failing to comply with the limitations of use may produce inaccurate results.
-
Despite the reliability of this test, there may be false negative and false positive results. If you are in any doubt about the result of your self-test, contact your doctor.
-
The self-test user should not make any medical decisions without consulting his/her doctor, whether the test is positive or negative.
-
Do not mix the components of differents batches of autotest COVID-VIRO ALL IN®.
-
The test instructions must be followed strictly to obtain an accurate result.
-
Do not use nasal sprays in the 12 hours before carrying out the test.
-
The diluent contains a solution with a preservative. If the solution comes into contact with the skin or eyes, rinse thoroughly with water.
-
AAZ-LMB accept no liability arising from any use, distribution or dispensing of the autotest COVID-VIRO ALL IN® or its parts that falls outside the indications and limitations of use stated in the current version of the instruction leaflet.
Legal notice
autotest COVID-VIRO ALL IN® is a self-test used to detect the COVID-19 virus by means of a nasal sample. This in vitro diagnostic device is for one-time use only and is designed fo use by non healthcare professionals in a private setting or with the help of healtcare professional.
Read the instructions in the user manual carefully. If you have any doubts about your results, contact you doctor.
Manufactured and distributed by: AAZ-LMB, 43 rue de Bellevue, 92100 Boulogne-Billancourt - France
Page reference: AAZ.AVA.24-A.
Date: novembre 8th 2021
Photo credit 123RF-157864381 - image : 123rf.com

 EN
EN FR
FR CZ
CZ PL
PL IT
IT DE
DE About this test
About this test

 Precautions and warnings
Precautions and warnings