autotest-sante.com: autotest de dépistage du VIH
autotest VIH®
In France, some 30,000 people are infected with HIV but unaware they are HIV*-positive. The risk of infection remains real.
Do the test at home!
autotest VIH® is a rapid HIV test you can do at home.
autotest VIH® is available in pharmacies (or on their websites) and can be purchased without a medical prescription.
autotest VIH® is reliable for the detection of an HIV infection that occurred at least 3 months ago using a blood sample taken from the tip of the finger.
- Test yourself in the privacy of your own home
- Easy to use (3 steps, 5 minutes to complete)
- Results in 15 minutes
* HIV is the virus responsible for AIDS

autotest VIH® is intended to be used by people who want to know their HIV status.
An estimated 150 000 people are living with HIV in France and 30 000 are unaware that they are HIV-positive. Not knowing about an infection can have serious health consequences and lead to further transmission of the virus. With 6 200 new HIV infections(1) recorded in France last year, the risk of infection remains real.
Certain groups of people are more at risk of being exposed to HIV because they are from a region with high prevalence or because of practices that put them at risk. These individuals are advised to get tested on a regular basis(1).
(1) Communiqué de presse du Ministère des Affaires sociales, de la Santé et des Droits des femmes. December 1st 2014.
(2) File « autotests de dépistage de l’infection par le VIH », Haute Autorité de Santé. March 2015.

There is currently no vaccine to prevent HIV infection. However, treatments exist for an infected individual that considerably reduce the risk of transmission.
autotest VIH® can serve as a complement to conventional testing services (3).
autotest VIH® is intended to provide individuals with a practical and confidential way to check their HIV status following a possible exposure to HIV that occurred more than 3 months ago.
In the event of a positive result, the test makes it possible for a person to seek medical care and benefit from treatment after having a confirmatory lab test. If HIV treatment is started earlier, life expectancy can be similar to the general population. In fact, the mortality of men infected with HIV and on antiretroviral therapy for more than three years is comparable to the mortality of men in the general population(4).
A negative result means that a person is probably not infected with HIV and can feel confident about his/her HIV status if the the most recent risk of exposure occurred at least 3 months ago.
(3) File « autotests de dépistage de l’infection par le VIH », Haute Autorité de Santé. March 2015.
(4) Prise en charge médicale des personnes infectées par le VIH, Pr. Patrick Yeni. Specialists, 2010. Page 52.
autotest VIH® is a blood test based on an immunochromatographic assay.
autotest VIH® does not require materials or equipment other than those included in the test kit. autotest VIH® can be used in private, at home.
With a drop of blood taken from the tip of the finger, autotest VIH® is able to highlight the presence of anti-HIV-1 and/or anti-HIV-2 antibodies.
autotest VIH® is available for sale in all pharmacies across France (and on their websites) and can be obtained without a medical prescription.
Speak to your pharmacist for more information.
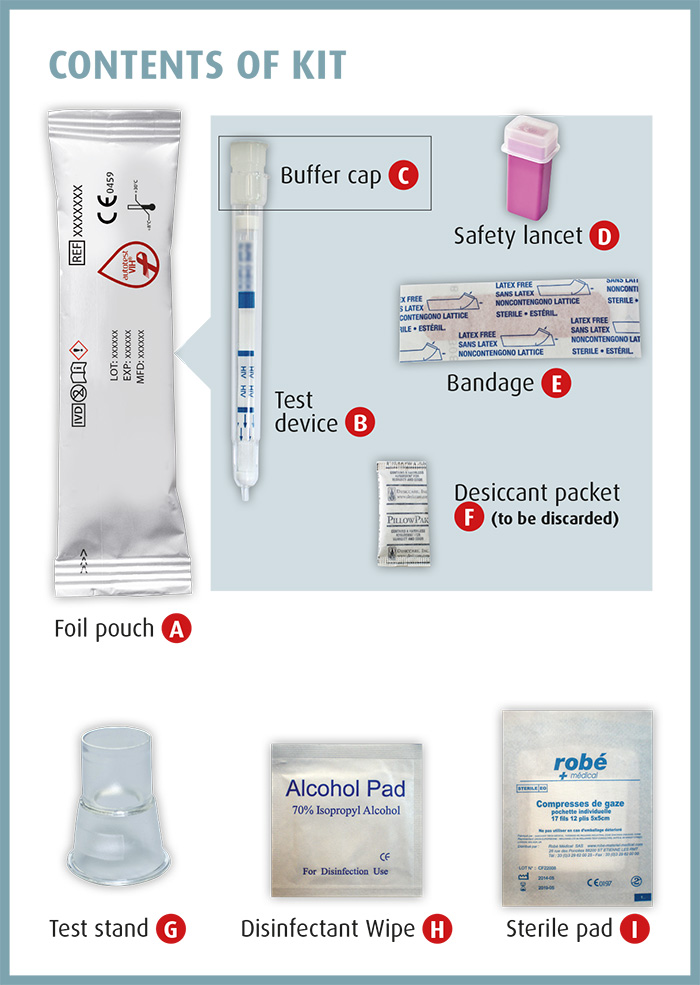
Every box contains:
- illustrated Instructions for use,
- a safety lancet to obtain a drop of blood,
- a test device with integrated sampling device that highlights whether anti-HIV-1 and/or anti-HIV-2 antibodies are present,
- a bandage.
It’s easy to get and read the result when using the autotest VIH® test. The test result appears as a line in just 15 minutes. If you have any doubts about the result, consult your doctor.
Packaging: single-use kit
Marqué CE 0459 (organisme notifié français LNE/G-MED)
Storage: between +8 and +30°C. Keep away from sunlight.
Type of sample: 2.5 microliters of capillary blood
Sensitivity (5): 100% of HIV-positive individuals obtained a correct result with this test.
Specificity(5): 99.8% of HIV-negative individuals obtained a correct result with this test.
Reliability: a practicability study of the handling of this test by laypeople showed that over 99.2% of participants obtained an interpretable result and over 98.1% correctly interpreted the result. Positive results were interpreted correctly in 100% of cases.
Interference: no significant interference has been observed upon examination of specimens that contain substances or that present medical conditions with the potential to affect the results of this test.
The window period (seroconversion) of the autotest VIH® test can be as long as three months meaning that HIV infections that occurred within the past three months might not be detected by this test.
(5) Studies of 503 people (sensitivity) and 2051 people (specificity) from the United States and the European Unio.
In relation to other types of testing, where does the autotest VIH® self-test fit in?
autotest VIH® is a rapid diagnostic orientation test. It is described as a unitary, subjective visual reading test that is easy to use and quickly provides a result (in under 20 minutes). It must be used with a sample of freshly collected capillary blood. Rapid tests are now able to detect both HIV-1 and HIV-2 antibodies. Some healthcare professionals and trained staff are able to use rapid tests in the course of their work. A negative result cannot be considered conclusive if the most recent risk of exposure to HIV occurred within the past 3 months. A positive result obtained using a rapid test must be confirmed by a laboratory test (4th generation Elisa test).
Who should do an HIV test and how often ?
Starting at what age can the autotest VIH® test be used?
What are the technical features that characterize the test?
How reliable are HIV self-tests?
Is autotest VIH® capable of detecting both HIV-1 and HIV-2?
Is the cost of purchasing an autotest VIH® test covered by Social Security?
How long do I have to wait after a risk of exposure to HIV before my autotest VIH® result can be considered reliable?
What are the optimal conditions for using the autotest VIH® test?
After opening the foil pouch containing the test device, how long can I wait before using the autotest VIH® test?
Can I use an autotest VIH® test if it has passed the expiry date printed on the packaging?
How can I be sure that I have handled the test properly and that there is no error that has affected the result?
What can I do if I have problems when using an autotest VIH® test?
I just bought my autotest VIH®, do I need additional materials or equipment?
Can the same autotest VIH® test be used more than once?
When the test is finished, what should I do with my used autotest VIH®?
What should I do if my test result is inconclusive?
What does does it mean if I get a positive result? What should I do?
What does does it mean if I get a negative result? What should I do ?
Can information about the autotest VIH® test be obtained by phone?
Where can I find a list and contact information for the various agencies that provide advice, support, medical care, and psychological counseling?
Can autotest VIH® be used to test for other sexually transmitted infections or for viral hepatitis?
If the result of my HIV self-test is negative, does this mean can I stop using condoms?
Can the fact that I am taking a medication have an effect on the autotest VIH® result?
Can I use the HIV self-test to monitor the effectiveness of my antiretroviral therapy?
Could someone else be infected handling a used HIV self-test?
- autotest VIH® must only be used with a fresh sample of capillary blood collected following the instructions as explained in this insert. DO NOT USE for serum or plasma specimens.
- Store the self-test in its original packaging in a cool, dry location between 8°C and 30°C. Keep away from sunlight
- autotest VIH® is for single-use only. DO NOT RE-USE.
- Do not open the foil pouch containing the self-test device until you are ready to perform the test.
- This self-test is not intended for use in the context of therapeutic follow-up with patients receiving antiretroviral therapy.
- Individuals who use this self-test should consult their doctor prior to making any medical decisions, regardless of whether the test result is negative or positive.
- False positive results (0.2% in studies of test specificity) or false negative results could be obtained in the following circumstances: exposure to HIV in the 3 months prior to using the test (window period), situations of advanced immunodeficiency or infection by a rare variant, and among HIV-positive people receiving antiretroviral therapy.
- Failure to store as directed or to respect the limits of use could cause the test result to be incorrect.
- Do not use the self-test if the packaging or foil pouch have been opened or damaged.
- Do not use if expiry date printed on the packaging has passed.
- If you have trouble understanding the package insert or instructions for use, please contact Sida Info Service immediately at 0 800 840 800 (24/7).
- In case of repeated inconclusive tests, please consult your doctor.
- Keep this self-test and the items that come with it away from children, the components included with autotest VIH® could be harmful if swallowed and could cause irritations.
- autotest VIH® is an additional form of HIV testing that can be used as a complement to other existing options. autotest VIH® is only able to detect HIV infection and cannot be used as a test for other sexually transmitted infections.
- AAZ-LMB expressly disclaims all liability for the use or distribution of autotest VIH® or any of its components, and for the consequences of inaccuracies or errors whether direct, indirect, incidental, or otherwise arising from any use of the autotest VIH® that fails to strictly follow the directions and limits of use as specified the applicable Instructions for Use.
Legal Information
Single-use in vitro diagnostic test with the CE Marking
Product denomination: autotest VIH® (ref: AS-VIH-001)
Purpose: autotest VIH® is intended for self-detection of HIV by a layperson in a private setting.
Correct use: this self-test is reliable for the detection of an HIV infection that occurred at least 3 months ago. In the event of repeated inconclusive results, consult your doctor.
Instructions: carefully read the Instructions for Use prior to using the test and consult your pharmacist if necessary.
Manufactured and distributed by
AAZ-LMB
43 rue de Bellevue
92100 Boulogne-Billancourt
FRANCE
Warning: In the event of any doubts about a test result, please consult your doctor.
This diagnostic medical device is a regulated health product that, in accordance with current regulations, bears the CE Marking.

 EN
EN FR
FR DK
DK CZ
CZ PL
PL LU
LU HU
HU CH - IT
CH - IT CH - FR
CH - FR CH - DE
CH - DE AT
AT RO
RO PT
PT ES
ES BE - DE
BE - DE BE - NL
BE - NL BE - FR
BE - FR NL
NL IT
IT DE
DE Who ?
Who ?




